What are TOC Cleaning Validation Swabs?
What are TOC Cleaning Validation Swabs?
TOC means Total organic carbon which analysis is a fast and effective analytical technique for cleaning validation in pharmaceutical manufacturing.
TOC swab is to control Microbial residue, can be use in HPLC, IMS or UV-VIS analysis
It is use to test water quality. Test the TOC.
TOC Swab < 50 PPb
• Material: knitted polyester,type is CM-PS713-TOC
• Head :Double layered
• Packing:Double packed ,20pcs/bag,50pcs/bag,100pcs/bag
• Washed by EDI pure water
• Low particle generation
• Good absorbent capacity
• Very good chemical resistance
• Autoclavable to ensure sampling under sterile conditions
• Shelf life :2 years
TOC cleaning validation sampling swab is a tool used for cleaning validation sampling, also known as TOC validation swab, TOC sampling stick, etc., is a very important tool in the biopharmaceutical industry, it is usually used to validate cleaners or disinfectants Whether the cleaning effect on the specific surface is satisfactory. In areas such as the medical, pharmaceutical and food industries, cleaning validation swabs are a vital task as they ensure the hygiene standards of the environment and equipment are met to safeguard the health and safety of the public.
What Are Cleaning Validation Swabs Made Of?
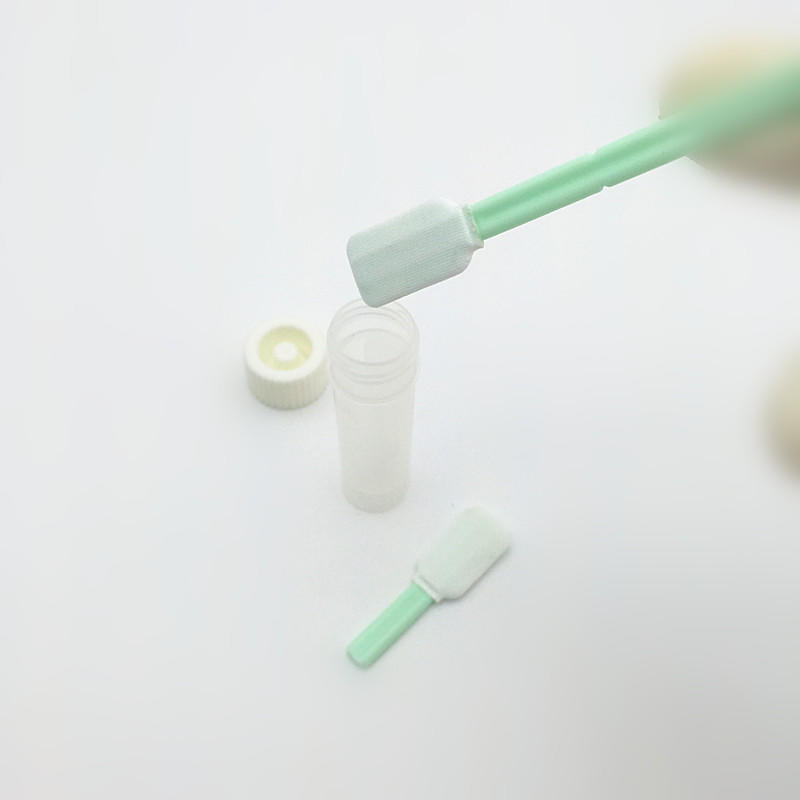
Cleaning validation swabs typically consist of a swab stick and a cloth head.
The tip is made of a special material that absorbs particles and organic matter left on the surface. When the cleaning verification swab is used, it will be soaked in the cleaner or disinfectant first, then gently brush the surface to be verified, and finally send the swab tip to the laboratory for analysis to determine whether the cleaning effect meets the standard.
The TOC sampling swab is designed with PP rod + medical cloth head. The material is soft, flat, not easy to defiber, and has good liquid absorption. The square head design has a wide contact range. The green rod has a groove design, which can be broken according to needs. Easy to operate, widely used in sampling for TOC detection or cleaning validation.
Why Cleaning Validation?
The concept of cleaning refers to the state where the total amount of various residues (including microorganisms and their metabolites) in the equipment is so low that it does not affect the prescribed curative effect, quality and safety of the next batch of products. Through effective cleaning, the previous batch of The substances remaining in the production equipment are reduced to the extent that they will not affect the curative effect, quality and safety of the next batch of products.
The purpose of cleaning verification is to prove that the established cleaning procedure can make the cleaning effect of the equipment clean through the test results, and the ultimate goal is to prevent drug contamination and cross-contamination.
Where To Buy High Quality Cleaning Validation Swabs?
If you are looking for high quality cleaning verification cotton swabs, then Cleanmo Technology Co., Ltd. will be your best choice. Our TOC Swab Stems and Tips are made from the highest quality raw materials, ensuring no contamination during use. The products have been strictly tested and verified to ensure that their quality and effect meet your needs. In addition, we also provide a variety of different types of cleaning verification swabs to meet the needs of different industries and application scenarios. Our products are cost-effective and affordable, which can save your business a lot of money.
CONTACT US
TEL : (+86)755-29455975
E-MAIL : info@cleanmo.com
FAX:(+86)755-61605135
OFFICE ADD : Room 908, 9/F., Qinchengda Building, Xin'an Street, Bao'an District, Shenzhen city, Guangdong Province P.R.China